 Also, the energies differ when carbon atoms are bound to one another through double/ triple bonds. It is 347kJ/mol for a C-C bond and 413 kJ/mol for a C-H bond. For instance, the bond energy of a C-C bond is different from the bond energy of a C-H bond. The positive sign is because it is the energy that is supplied to break the chemical bonds. It is to be noted that the bond energy is always a positive value. The unit of bond energy is kcal/ mol or kilojoule /mol, abbreviated as kJ/mol. Now that you know what bond energies are, it is important to understand the factors affecting the bond energies of various atoms and molecules. The total energy will be divided by four, i.e., the total number of similar bonds in the molecule. Thus, the bond energy of methane’s carbon-hydrogen bond can be pictured as the change in enthalpy associated with the breakage of a CH 4 molecule into 1 carbon atom and 4 hydrogen atoms. BE(C-H), in the last equation, represents the bond energy of the C-H bond in the methane molecule, which is calculated by taking the average of the four energies mentioned above.Bond Dissociation E 4 is the energy needed to break the only C-H bond in the CH molecule.Bond Dissociation E 3 is the energy required to break a C-H bond in the CH 2.Similarly, Bond Dissociation E 2 is the energy needed to break a C–H bond in the CH 3.Bond Dissociation E 1 denotes the amount of energy needed to break one C–H bond in the CH 4.It can be calculated as follows.īE(C-H) = (Bond Dissociation E 1 + Bond Dissociation E 2 + Bond Dissociation E 3 + Bond Dissociation DE 4)/4 The bond energy of a C-H (carbon-hydrogen) bond in a methane molecule will be equal to the average of the bond dissociation enthalpies of every carbon-hydrogen bond. The following structural diagram of methane will give you a complete idea of the chemical bonds in a molecule. This average is for all the individual bond dissociation enthalpies (energies) of the bonds.įor instance, suppose you consider a methane molecule. Understanding Bond Energies with Exampleįor a clear understanding of the concept, it must be remembered that the bond energy of a chemical bond in a chemical compound is the average value. 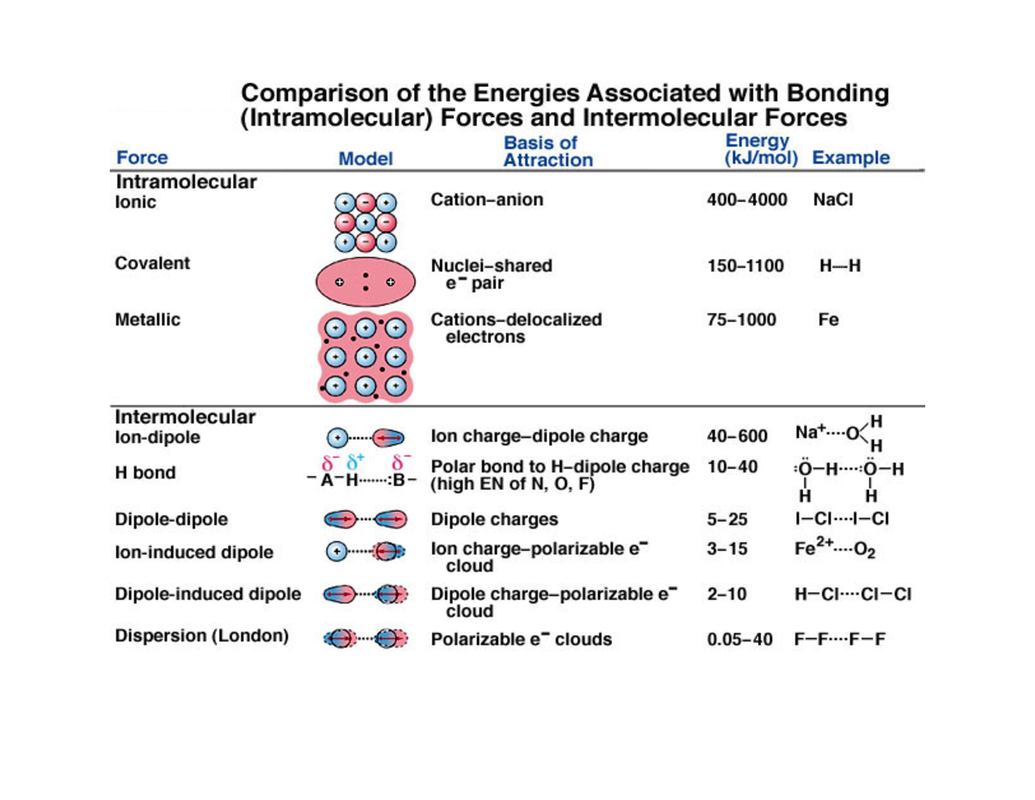
Thus, the bond energies of chemical bonds in compounds are the average amount of energy needed to break such chemical bonds. It must be noted that the enthalpies are taken in the gaseous state. 
“The value obtained as an average of the bond dissociation enthalpies (energies) of all chemical bonds of a particular type for a given compound” It is the energy quantity that gives an understanding of the strength of a chemical bond.īond Energy Definition: The term bond energy can be defined as follows: What is Bond Energy?īond energy is also referred to as average bond enthalpy or bond enthalpy. The following article thoroughly studies bond energies and how they vary for different elements and compounds. And that is what bond energy definition is all about. 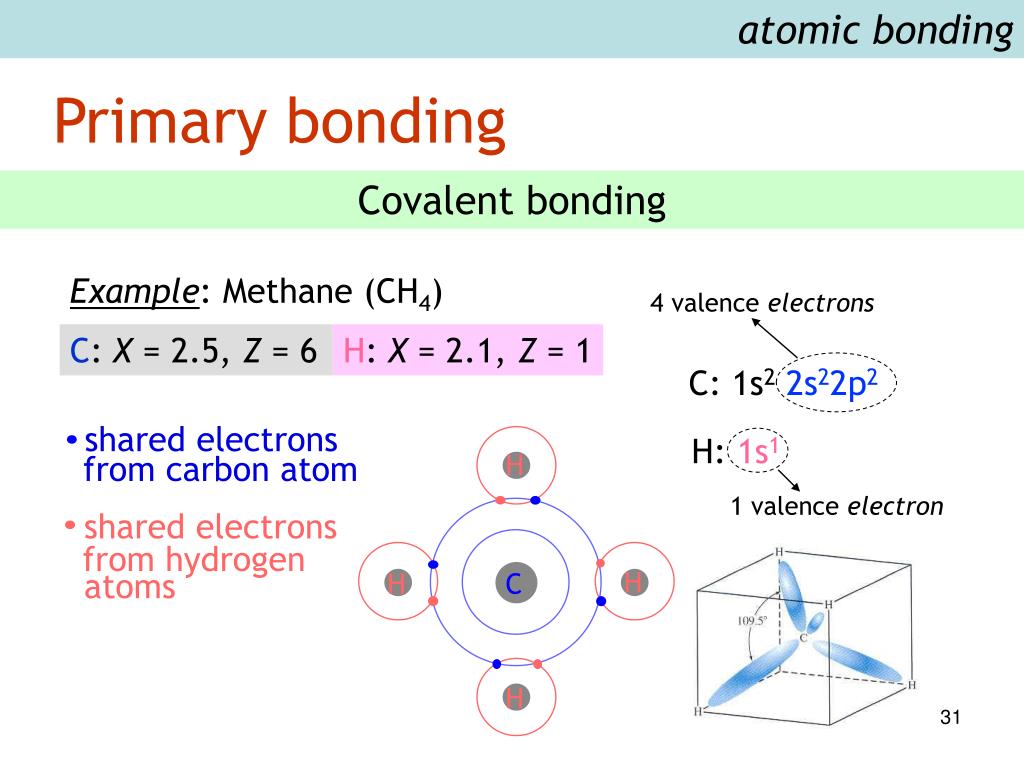
This definition leaves you wondering how strong is the force of attraction. But what is a bond? The strong force of attraction between ions, atoms, or molecules facilitates the formation of chemical compounds. Atoms in a molecule are connected to one another by bonds. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
